 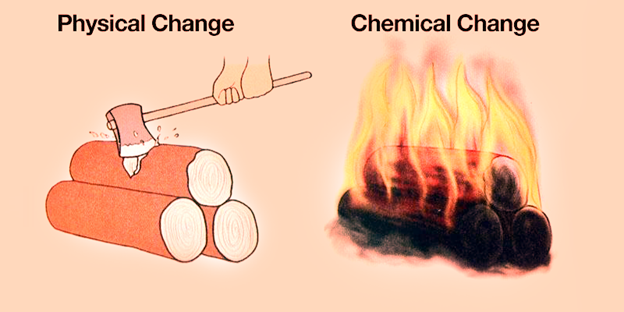
As the rust forms on the surface of the iron, it flakes off to expose more iron, which will continue to rust. 3: Rust (iron oxide) forms on an unprotected iron surface. See how to distinguish between physical and chemical changes with an alloy example. Physical changes are changes of shape, texture, or state of a material, while chemical changes are changes of molecular structure or composition. 
The starting materials may not smell at all but as you mix these materials you may end up with a bad odor or a pleasant one. When exposed to air, an object made of iron will eventually begin to rust (see figure below). Learn the difference between physical and chemical changes in matter, and how to identify them with examples. You may start with two colorless solutions but when they are mixed you might see a bright purple color. Note: reactions that produce heat are known as exothermic reactions whereas reactions that absorb heat are known as endothermic reactions. The temperature could go up (gets hotter) or the temperature could go down (gets colder). *The reaction produced a change in temperature. Examples of chemical changes would be the reaction of iron with air (rusting} or the reaction of a metal and acid.Ĭertain observations will indicate that a chemical change has occurred. See examples of physical and chemical changes in everyday life, such as melting, burning, rusting, and dissolving. This new substance has a different chemical composition than the starting materials. Learn the difference between physical and chemical changes in matter, how to identify them, and why they are important. A chemical change is a change in which something new is formed (irreversible). The starting materials change into an entirely different substance or substances. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
